|
4/1/2023 0 Comments Complexed ion bonding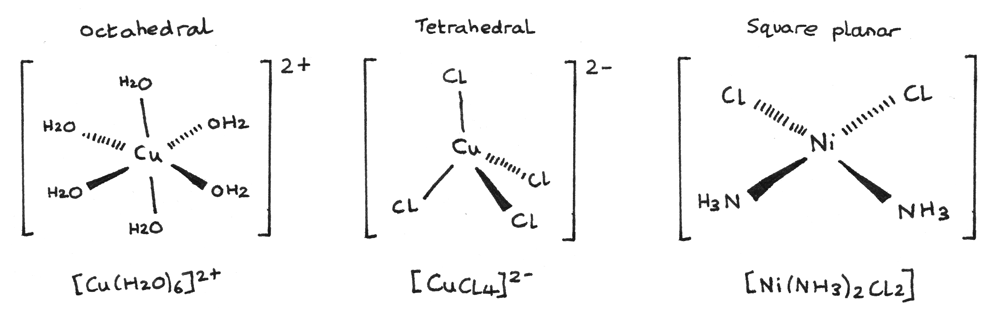 
Other common ligands include carbon monoxide, acetylacetonate, and ethylenediamine, as well as metal ions, such as chloride, cyanide, and nitrate. Ligands can also be more complex molecules or ions, such as organic compounds or metal ions. Some common ligands include water, ammonia, and other simple molecules that can easily donate a pair of electrons to form a bond with the central atom or ion. This allows the ligand to coordinate (or bind) to the central atom or ion, creating a coordination complex. Ligands are typically Lewis bases, which means that they have at least one pair of non-bonded electrons that they can use to form a bond with the central atom or ion. Ligands are often used in coordination chemistry to form coordination compounds, which are molecules or ions that consist of a central atom or ion bonded to one or more ligands. In chemistry, a ligand is a molecule or ion that binds to a central atom or ion to form a coordination complex. Overall, complex ions are an important part of coordination chemistry, and they play a key role in many chemical processes and applications. They are also used in a variety of applications, such as in the synthesis of drugs and other compounds, in the separation and purification of molecules, and in the detection of specific ions or molecules. The stability of a complex ion can also depend on the nature of the bonds between the central atom or ion and the ligands, as well as the interaction between the ligands themselves.Ĭomplex ions are important in many fields of chemistry, including inorganic chemistry, biochemistry, and analytical chemistry. For example, the color of a complex ion can depend on the type of ligands it contains and the way they are arranged around the central atom or ion. The properties of a complex ion depend on the identity of the central atom or ion and the ligands it is bonded to. Often, chemical complexes have an ionic charge and thus form complex ions. Complex ions are also known as coordination complexes. Conversely, central metal ions count as Lewis acids because they accept pairs of electrons. Since ligands donate pairs of electrons, they are classified as Lewis bases. Chemists define “ligand” as a molecule capable of donating a pair of electrons to form a coordinate covalent bond. In inorganic chemistry, a “complex” describes a structure involving a central metal ion with coordinate covalent bonds to one or more ligands. In this article, we learn about complex ions in chemistry and their ligands, including the various types of complexes, the importance of coordination number, and the chemistry of ligands including ligand substitution reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
